The antimicrobial resistance crisis: stopping mission impossible
The consequences of the AMR crisis for modern medicine
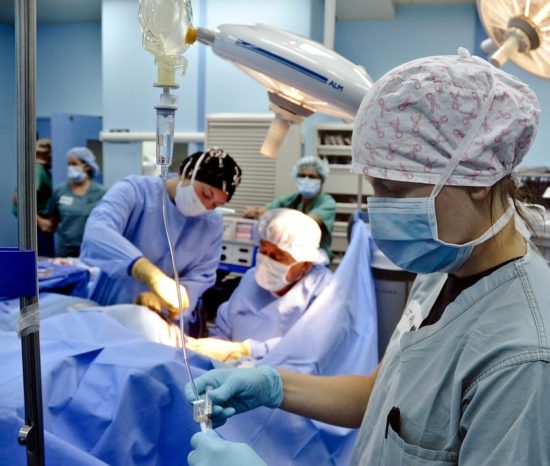
All achievements of modern medicine are only possible through the application of medical devices (MD). This is especially true for class three invasive medical devices according to the European Union’s medical device regulations (MDR). Annually, around 180,000 pacemakers, 260,000 cardiovascular stents, 200,000 hernia meshes, 365,000 hip and knee endoprostheses are implanted into patients in Germany. Furthermore, millions of central venous catheters, ports, urinary catheters, and wound dressings for example are used invasively. All of these implants are made of biomaterials with medical grade approval. Amongst them are different classes of materials, such as metal alloys, ceramics and polymers. Although these biomaterials are mainly described as ‘inert’ (they do not harm the host’s biology), or bio-active to support the host’s biology in different ways, they still remain as foreign materials. These foreign materials possess the disadvantage that bacteria and commensals of the host and pathogens interact with these biomaterials in a severely dangerous manner. Bacteria can form biofilms on the outer and inner surfaces of such biomaterials, often avoiding the host’s immune defence or applied antibiotics if the patient is suffering from an infection.
AMR NEWS
Your Biweekly Source for Global AMR Insights!
Stay informed with the essential newsletter that brings together all the latest One Health news on antimicrobial resistance. Delivered straight to your inbox every two weeks, AMR NEWS provides a curated selection of international insights, key publications, and the latest updates in the fight against AMR.
Don’t miss out on staying ahead in the global AMR movement—subscribe now!