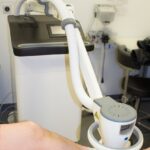
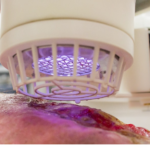
SteriPlas – cold argon plasma antibacterial
Adtec's SteriPlas has proven clinical efficacy in infection management of wounds, diabetic foot ulcers and surgical site infections. With a physical mode of action, can be used as a topical alternative to antibiotics to help in the fight against AMR.
Clinical Trials conducted using Adtec microwave argon plasma results include a 73.5% reduction in bacterial load with standard of care plus plasma treatment compared to a 50% reduction with standard of care alone. This represents a 47% increase in bacterial load reduction with SOC and plasma compared to SOC. All 24 patients received standard wound care to all wounds; 22 patients also received systemic antibiotics (92%).
- Antimicrobial compound/strategy
- Infection prevention
- Antimicrobial stewardship
- Microbial diagnostics
- Removal antibiotics/bacteria
Microorganisms:
- Bacteria
- Fungi
- Viruses
- Yeasts
- Parasites
Application:
- Human
- Veterinary
- AgriFood
- Environmental
- Other
Development stage:
- Marketed product
- Research
- Development
- Validation
- Market entry
- Company
- Academia
- Institute
- NGO
- Government
Partnering:
- Joint Venture
- License
- Co-develop
- Outsource
- Sell
Funding organisation:
- OTHER / NA
- CARB-X
- FIND
- GARDP
- REPAIR
Infectious disease area:
- SSTI
- SSI
- UTI
- STI
- BSI
- RTI
- GII
- CNSI
- IAI
Geographic origin:
- Eurasia
- North America
- South America
- Africa
- Oceania
Joint venture and open to investment to fund FDA approval.
Adtec Healthcare Limited is a UK company commercializing innovative cold plasma technology.
AHL designs and manufactures the SteriPlas medical device (ISO13485: 2016 QMS accredited). SteriPlas infection management argon gas plasma technology is antibacterial with proven clinical and safety efficacy in treatment of biofilms, wounds, surgical site infections and medical dermatology conditions. As a topical antibacterial with a physical mode of action, bacterial resistance is unlikely to develop. It can therefore be considered as an alternative therapeutic to antibiotics helping to combat antibacterial resistance.
SteriPlas technology has proven efficacy to treat Actinic Keratosis and other medical dermatological conditions.
(www.adtechealthcare.com).
Cold argon plasma for treating bacterial skin conditions in medical dermatology.